  
7 In addition, the randomized, placebo-controlled, phase 3 KEYNOTE-407 study showed significantly improved OS and PFS with pembrolizumab plus carboplatin and paclitaxel or nab-paclitaxel versus placebo plus chemotherapy in patients with stage IV squamous NSCLC. 6 The randomized, placebo-controlled, phase 3 KEYNOTE-189 study, a larger confirmatory clinical trial, demonstrated significantly improved overall survival (OS) and progression-free survival (PFS) with pembrolizumab plus pemetrexed and carboplatin or cisplatin versus placebo plus chemotherapy in patients with metastatic nonsquamous NSCLC without sensitizing EGFR or ALK alterations. In cohort G of the multicohort, phase 1/2 KEYNOTE-021 study, a significantly higher objective response rate (ORR) was observed with pembrolizumab plus pemetrexed and carboplatin versus chemotherapy alone in patients with previously untreated advanced nonsquamous NSCLC and no sensitizing EGFR or ALK alterations. 4, 5 Chemotherapy may also enhance the antitumor activity of immunotherapy 4 and thus increase the likelihood of a clinical benefit with pembrolizumab, regardless of tumor PD-L1 expression. On the basis of results from KEYNOTE-001, patients with PD-L1‒negative tumors (PD-L1 TPS < 1%) were not included in these studies, and they are not eligible for pembrolizumab monotherapy.Ĭhemotherapy agents have shown immunomodulatory properties, including directly and indirectly stimulating immune responses and increasing tumor immunogenicity. 1 Results from the phase 3 studies KEYNOTE-024 and KEYNOTE-042 for patients with PD-L1 expression on ≥50% of tumor cells (tumor proportion score ≥ 50%) 2 and for patients with a PD-L1 TPS ≥ 1%, 3 respectively, subsequently established pembrolizumab monotherapy as a first-line standard of care for patients with PD-L1‒positive advanced NSCLC and no EGFR or ALK genomic tumor aberrations. The anti–programmed death 1 (PD-1) monoclonal antibody pembrolizumab first showed increasing clinical efficacy with higher levels of programmed death ligand 1 (PD-L1) expression inpatients with advanced non–small cell lung cancer (NSCLC) in the phase 1b KEYNOTE-001 study. Pembrolizumab plus chemotherapy demonstrated response and survival improvements with manageable safety in comparison with chemotherapy alone in PD-L1‒negative advanced/metastatic NSCLC, and it is a standard-of-care first-line therapy for patients with advanced NSCLC, regardless of PD-L1 expression. Adverse events (AEs) were experienced by 99.2% of the patients who received pembrolizumab plus chemotherapy and by 98.9% of the patients who received chemotherapy alone, with grade 3 or higher AEs occurring in 71.4% and 72.0%, respectively immune-mediated AEs and infusion reactions were experienced by 29.0% and 12.4%, respectively. 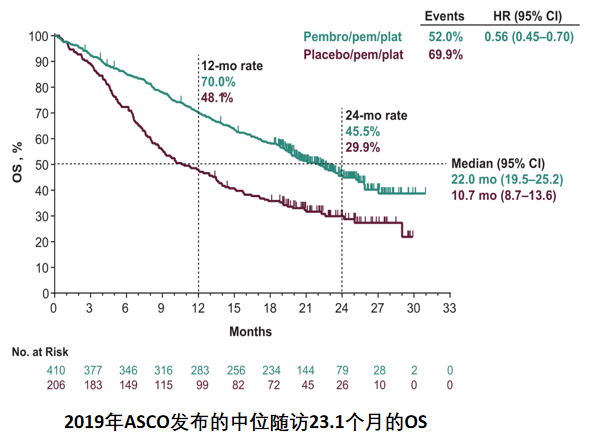
Sixteen patients in the pembrolizumab plus chemotherapy arm completed 2 years of treatment the objective response rate was 87.5% (95% CI, 61.7%-98.4%), and the 3-year OS rate was 100%. Pembrolizumab plus chemotherapy improved overall survival (OS hazard ratio, 0.63 95% CI, 0.50-0.79) and progression-free survival (HR, 0.68 95% CI, 0.56-0.83) over chemotherapy. The median time from randomization to the data cutoff was 28.0 months (range, 14.7-55.4 months). Resultsįour hundred forty-four of the 1328 patients (33.4%) who were enrolled across the 3 trials had PD-L1‒negative tumors (256 on pembrolizumab plus chemotherapy and 188 on chemotherapy alone ). 
No α was assigned to this descriptive, exploratory analysis. Responses were assessed according to Response Evaluation Criteria in Solid Tumors version 1.1 by blinded, independent, central review. Treatment comprised pembrolizumab plus chemotherapy (pemetrexed and platinum for nonsquamous histology and carboplatin and paclitaxel/nab-paclitaxel for squamous histology) or chemotherapy alone. Individual patient data were pooled from KEYNOTE-021 cohort G (nonsquamous NCT02039674), KEYNOTE-189 (nonsquamous NCT02578680 and NCT03950674), and KEYNOTE-407 (squamous NCT02775435). This study pooled data from 3 randomized controlled trials to evaluate outcomes with pembrolizumab plus chemotherapy versus chemotherapy alone in patients with advanced/metastatic NSCLC negative for PD-L1 (ie, a tumor proportion score < 1%). Pembrolizumab plus platinum-based chemotherapy has demonstrated improved clinical outcomes over chemotherapy alone in patients with previously untreated advanced/metastatic non–small cell lung cancer (NSCLC), regardless of tumor programmed death ligand 1 (PD-L1) expression. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
